- Volumes 84-95 (2024)
-
Volumes 72-83 (2023)
-
Volume 83
Pages 1-258 (December 2023)
-
Volume 82
Pages 1-204 (November 2023)
-
Volume 81
Pages 1-188 (October 2023)
-
Volume 80
Pages 1-202 (September 2023)
-
Volume 79
Pages 1-172 (August 2023)
-
Volume 78
Pages 1-146 (July 2023)
-
Volume 77
Pages 1-152 (June 2023)
-
Volume 76
Pages 1-176 (May 2023)
-
Volume 75
Pages 1-228 (April 2023)
-
Volume 74
Pages 1-200 (March 2023)
-
Volume 73
Pages 1-138 (February 2023)
-
Volume 72
Pages 1-144 (January 2023)
-
Volume 83
-
Volumes 60-71 (2022)
-
Volume 71
Pages 1-108 (December 2022)
-
Volume 70
Pages 1-106 (November 2022)
-
Volume 69
Pages 1-122 (October 2022)
-
Volume 68
Pages 1-124 (September 2022)
-
Volume 67
Pages 1-102 (August 2022)
-
Volume 66
Pages 1-112 (July 2022)
-
Volume 65
Pages 1-138 (June 2022)
-
Volume 64
Pages 1-186 (May 2022)
-
Volume 63
Pages 1-124 (April 2022)
-
Volume 62
Pages 1-104 (March 2022)
-
Volume 61
Pages 1-120 (February 2022)
-
Volume 60
Pages 1-124 (January 2022)
-
Volume 71
- Volumes 54-59 (2021)
- Volumes 48-53 (2020)
- Volumes 42-47 (2019)
- Volumes 36-41 (2018)
- Volumes 30-35 (2017)
- Volumes 24-29 (2016)
- Volumes 18-23 (2015)
- Volumes 12-17 (2014)
- Volume 11 (2013)
- Volume 10 (2012)
- Volume 9 (2011)
- Volume 8 (2010)
- Volume 7 (2009)
- Volume 6 (2008)
- Volume 5 (2007)
- Volume 4 (2006)
- Volume 3 (2005)
- Volume 2 (2004)
- Volume 1 (2003)
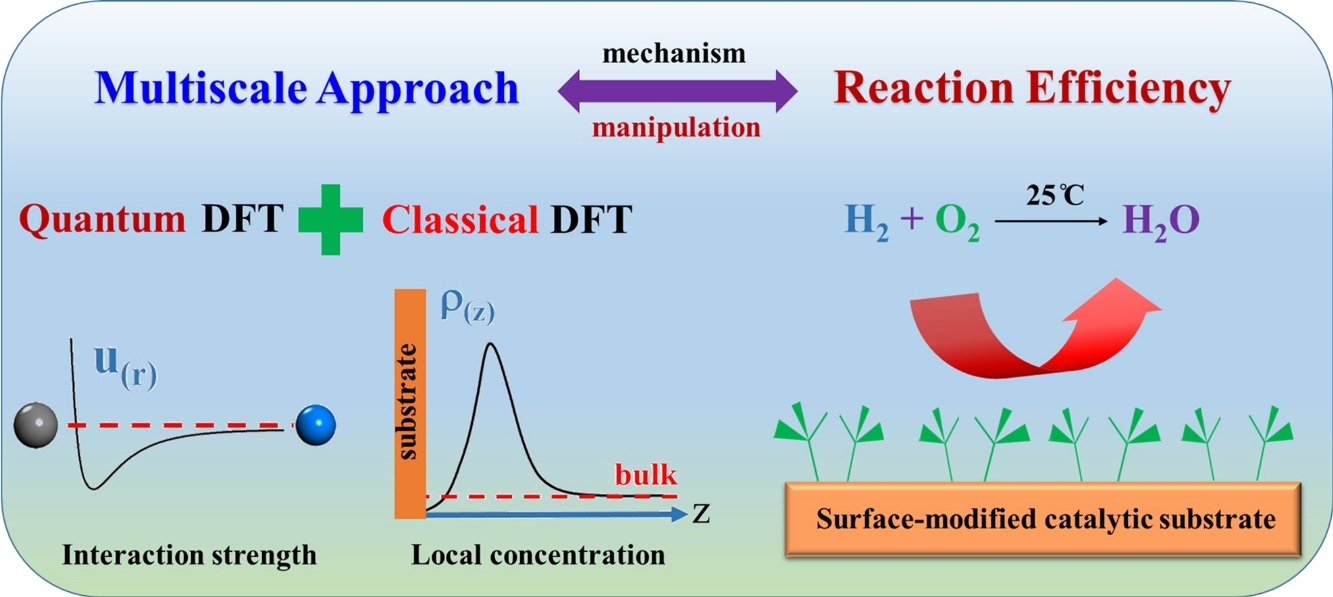
• A multiscale approach was developed towards the surface catalytic reaction efficiency.
• Hydrogen off-gas oxidation on different surface-modified catalytic substrates was investigated.
• Optimum surface modifications for efficient reaction is identified and compared with experiment.
A catalytic micro-reactor for converting hydrogen off-gas into water was recently developed, through which the conversion efficiency of hydrogen gas was greatly improved by hydrophobic modification of the catalytic substrate. Herein, a hybrid theoretical method is reported that combines density functional theory (DFT) on both the quantum and molecular scales. This method allows the microscopic study of the mechanism by which the surface catalytic reaction can be manipulated. Specifically, quantum DFT calculations are performed to quantify the molecular interaction between the catalytic substrate and reagent or product. Classical DFT investigations are subsequently carried out to determine the local concentrations of reagents near catalytic sites subject to different surface coating conditions. Finally, the reaction efficiency is determined from the local concentrations based on collision theory. This multiscale method provides molecular insight for quantifying the effect of catalytic surface modification on the reaction efficiency. The method reveals that an optimal surface hydrophobic modification can promote the densities of reagents near the substrate, while depleting the produced water. These two factors promote the conversion efficiency. The exclusion of produced water from the catalytic substrate is affected more by the degree of polymer grafting than by the chain length of hydrophobic polymer moieties.