- Volumes 84-95 (2024)
-
Volumes 72-83 (2023)
-
Volume 83
Pages 1-258 (December 2023)
-
Volume 82
Pages 1-204 (November 2023)
-
Volume 81
Pages 1-188 (October 2023)
-
Volume 80
Pages 1-202 (September 2023)
-
Volume 79
Pages 1-172 (August 2023)
-
Volume 78
Pages 1-146 (July 2023)
-
Volume 77
Pages 1-152 (June 2023)
-
Volume 76
Pages 1-176 (May 2023)
-
Volume 75
Pages 1-228 (April 2023)
-
Volume 74
Pages 1-200 (March 2023)
-
Volume 73
Pages 1-138 (February 2023)
-
Volume 72
Pages 1-144 (January 2023)
-
Volume 83
-
Volumes 60-71 (2022)
-
Volume 71
Pages 1-108 (December 2022)
-
Volume 70
Pages 1-106 (November 2022)
-
Volume 69
Pages 1-122 (October 2022)
-
Volume 68
Pages 1-124 (September 2022)
-
Volume 67
Pages 1-102 (August 2022)
-
Volume 66
Pages 1-112 (July 2022)
-
Volume 65
Pages 1-138 (June 2022)
-
Volume 64
Pages 1-186 (May 2022)
-
Volume 63
Pages 1-124 (April 2022)
-
Volume 62
Pages 1-104 (March 2022)
-
Volume 61
Pages 1-120 (February 2022)
-
Volume 60
Pages 1-124 (January 2022)
-
Volume 71
- Volumes 54-59 (2021)
- Volumes 48-53 (2020)
- Volumes 42-47 (2019)
- Volumes 36-41 (2018)
- Volumes 30-35 (2017)
- Volumes 24-29 (2016)
- Volumes 18-23 (2015)
- Volumes 12-17 (2014)
- Volume 11 (2013)
- Volume 10 (2012)
- Volume 9 (2011)
- Volume 8 (2010)
- Volume 7 (2009)
- Volume 6 (2008)
- Volume 5 (2007)
- Volume 4 (2006)
- Volume 3 (2005)
- Volume 2 (2004)
- Volume 1 (2003)
• CO2 methanation using renewable energy is a promising process for CO2 recycling.
• Model of fluidized-bed reactor for CO2 methanation.
• Characterized reaction kinetics of the NiMgW catalyst for model validation.
• Sensitivity analysis to determine effects of operating parameters on reactor performance.
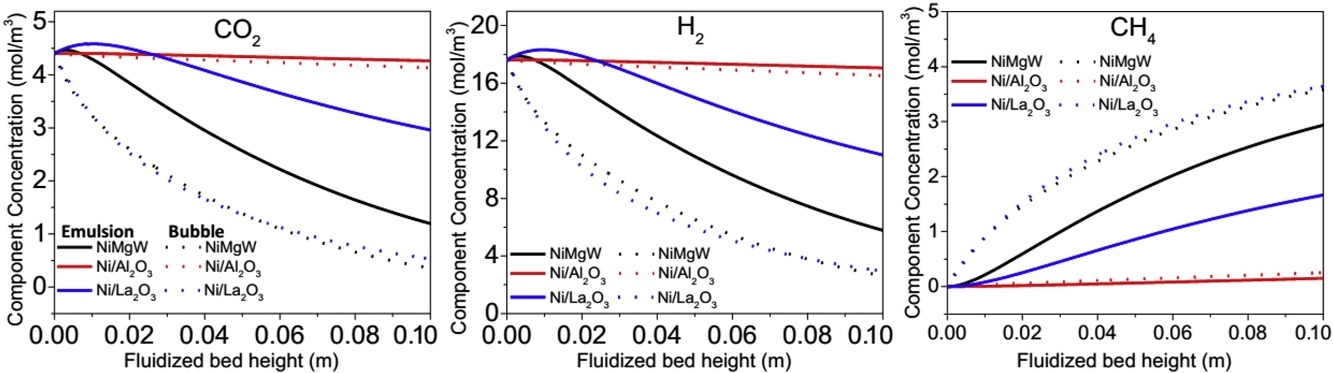
The reduction of carbon dioxide to methane by hydrogen ("CO2 methanation") using renewable energy is a promising process for recycling CO2. Better catalysts and better reactors are both required for the practical application of CO2 methanation. This study examines how the operating parameters affect CO2 methanation in a highly efficient fluidized-bed reactor. We first measured the kinetics of the CO2 methanation reaction using an NiMgW catalyst, which has been reported to exhibit superior catalytic performance. We then developed a fluidized-bed reactor model based on an earlier model for CO2 methanation. The fluidized bed model indicated that the NiMgW was indeed superior to two other previously studied catalysts in terms of faster conversion of reactants and higher concentrations of product CH4 throughout the reactor. The overall rate of production of CH4 increased with temperature and H2/CO2 ratio and decreased as the inlet reactant flow rate, catalyst particle diameter, and catalyst particle sphericity increased.