- Volumes 84-95 (2024)
-
Volumes 72-83 (2023)
-
Volume 83
Pages 1-258 (December 2023)
-
Volume 82
Pages 1-204 (November 2023)
-
Volume 81
Pages 1-188 (October 2023)
-
Volume 80
Pages 1-202 (September 2023)
-
Volume 79
Pages 1-172 (August 2023)
-
Volume 78
Pages 1-146 (July 2023)
-
Volume 77
Pages 1-152 (June 2023)
-
Volume 76
Pages 1-176 (May 2023)
-
Volume 75
Pages 1-228 (April 2023)
-
Volume 74
Pages 1-200 (March 2023)
-
Volume 73
Pages 1-138 (February 2023)
-
Volume 72
Pages 1-144 (January 2023)
-
Volume 83
-
Volumes 60-71 (2022)
-
Volume 71
Pages 1-108 (December 2022)
-
Volume 70
Pages 1-106 (November 2022)
-
Volume 69
Pages 1-122 (October 2022)
-
Volume 68
Pages 1-124 (September 2022)
-
Volume 67
Pages 1-102 (August 2022)
-
Volume 66
Pages 1-112 (July 2022)
-
Volume 65
Pages 1-138 (June 2022)
-
Volume 64
Pages 1-186 (May 2022)
-
Volume 63
Pages 1-124 (April 2022)
-
Volume 62
Pages 1-104 (March 2022)
-
Volume 61
Pages 1-120 (February 2022)
-
Volume 60
Pages 1-124 (January 2022)
-
Volume 71
- Volumes 54-59 (2021)
- Volumes 48-53 (2020)
- Volumes 42-47 (2019)
- Volumes 36-41 (2018)
- Volumes 30-35 (2017)
- Volumes 24-29 (2016)
- Volumes 18-23 (2015)
- Volumes 12-17 (2014)
- Volume 11 (2013)
- Volume 10 (2012)
- Volume 9 (2011)
- Volume 8 (2010)
- Volume 7 (2009)
- Volume 6 (2008)
- Volume 5 (2007)
- Volume 4 (2006)
- Volume 3 (2005)
- Volume 2 (2004)
- Volume 1 (2003)
• Process of forming a caffeine crystal suspension was investigated.
• Weak intermolecular interactions are responsible for viscosity.
• Effect of temperature and supersaturation ratio on viscosity was studied.
• Viscous mechanism of caffeine crystallization solutions was proposed.
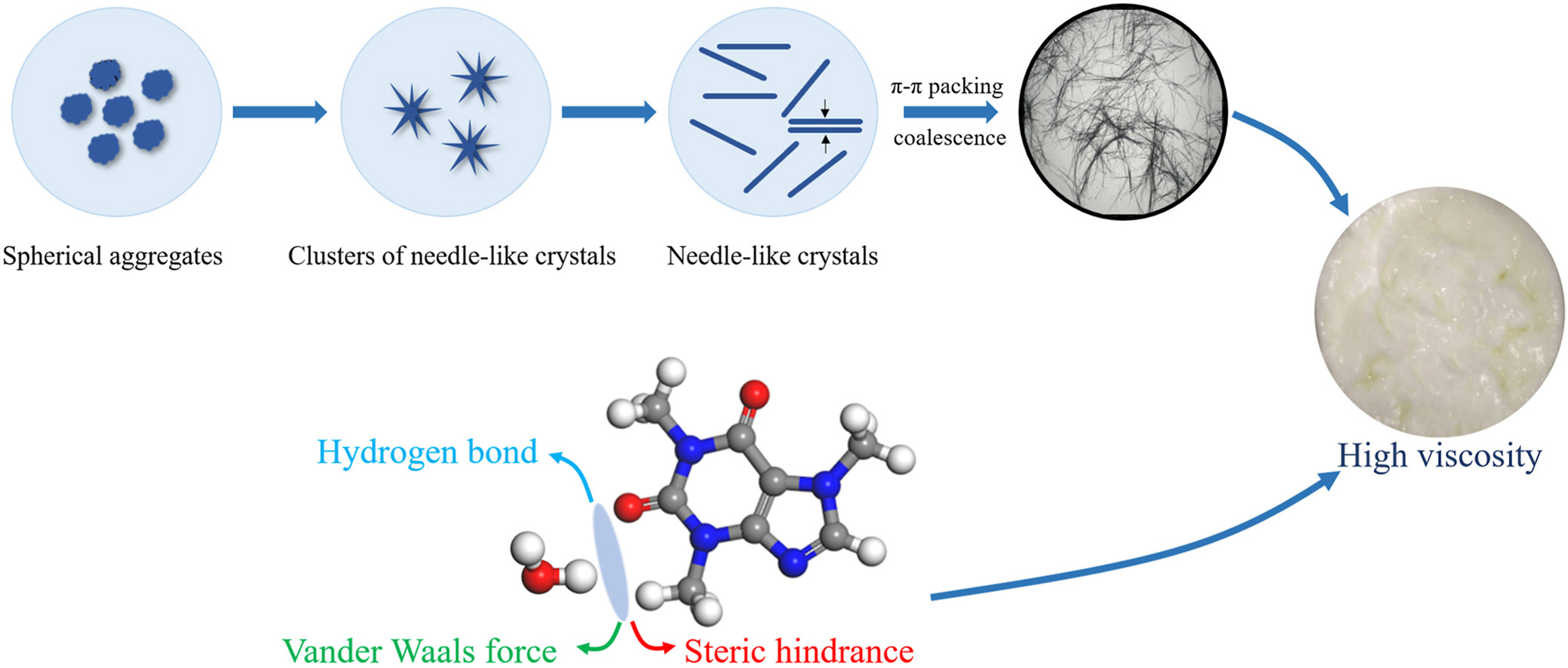
The solid-liquid viscous system formed by high viscosity crystallization solution impacts the flow and separation performance. Therefore, it is very important to study the viscosity mechanism to improve viscosity and regulate crystallization, ensuring a seamless production process. Herein, the viscosity of crystallization solution was taken as the measurement parameter of caffeine as a model drug. We investigated the viscosity mechanism of caffeine crystallization solutions by combining experiment and simulation. The results indicated that the weak interactions between caffeine and water result in increased viscosity of the caffeine crystallization solutions. Moreover, caffeine crystals possess elongated needle-like shapes, featuring a substantial specific surface area. Additionally, there is π-π stacking occurring between the (2 0 0) crystal face and (1 1 0) crystal face, effectively fostering coalescence of the crystals towards the radial side of the crystal along its elongated axis, resulting in a more viscous crystallization system. The results contribute to comprehending the viscosity mechanism of crystal systems and provide theoretical foundation to enhance engineering efficiency in crystallization.