-
Volumes 84-95 (2024)
-
Volume 94
Pages 1-400 (November 2024)
-
Volume 93
Pages 1-376 (October 2024)
-
Volume 92
Pages 1-316 (September 2024)
-
Volume 91
Pages 1-378 (August 2024)
-
Volume 90
Pages 1-580 (July 2024)
-
Volume 89
Pages 1-278 (June 2024)
-
Volume 88
Pages 1-350 (May 2024)
-
Volume 87
Pages 1-338 (April 2024)
-
Volume 86
Pages 1-312 (March 2024)
-
Volume 85
Pages 1-334 (February 2024)
-
Volume 84
Pages 1-308 (January 2024)
-
Volume 94
-
Volumes 72-83 (2023)
-
Volume 83
Pages 1-258 (December 2023)
-
Volume 82
Pages 1-204 (November 2023)
-
Volume 81
Pages 1-188 (October 2023)
-
Volume 80
Pages 1-202 (September 2023)
-
Volume 79
Pages 1-172 (August 2023)
-
Volume 78
Pages 1-146 (July 2023)
-
Volume 77
Pages 1-152 (June 2023)
-
Volume 76
Pages 1-176 (May 2023)
-
Volume 75
Pages 1-228 (April 2023)
-
Volume 74
Pages 1-200 (March 2023)
-
Volume 73
Pages 1-138 (February 2023)
-
Volume 72
Pages 1-144 (January 2023)
-
Volume 83
-
Volumes 60-71 (2022)
-
Volume 71
Pages 1-108 (December 2022)
-
Volume 70
Pages 1-106 (November 2022)
-
Volume 69
Pages 1-122 (October 2022)
-
Volume 68
Pages 1-124 (September 2022)
-
Volume 67
Pages 1-102 (August 2022)
-
Volume 66
Pages 1-112 (July 2022)
-
Volume 65
Pages 1-138 (June 2022)
-
Volume 64
Pages 1-186 (May 2022)
-
Volume 63
Pages 1-124 (April 2022)
-
Volume 62
Pages 1-104 (March 2022)
-
Volume 61
Pages 1-120 (February 2022)
-
Volume 60
Pages 1-124 (January 2022)
-
Volume 71
- Volumes 54-59 (2021)
- Volumes 48-53 (2020)
- Volumes 42-47 (2019)
- Volumes 36-41 (2018)
- Volumes 30-35 (2017)
- Volumes 24-29 (2016)
- Volumes 18-23 (2015)
- Volumes 12-17 (2014)
- Volume 11 (2013)
- Volume 10 (2012)
- Volume 9 (2011)
- Volume 8 (2010)
- Volume 7 (2009)
- Volume 6 (2008)
- Volume 5 (2007)
- Volume 4 (2006)
- Volume 3 (2005)
- Volume 2 (2004)
- Volume 1 (2003)
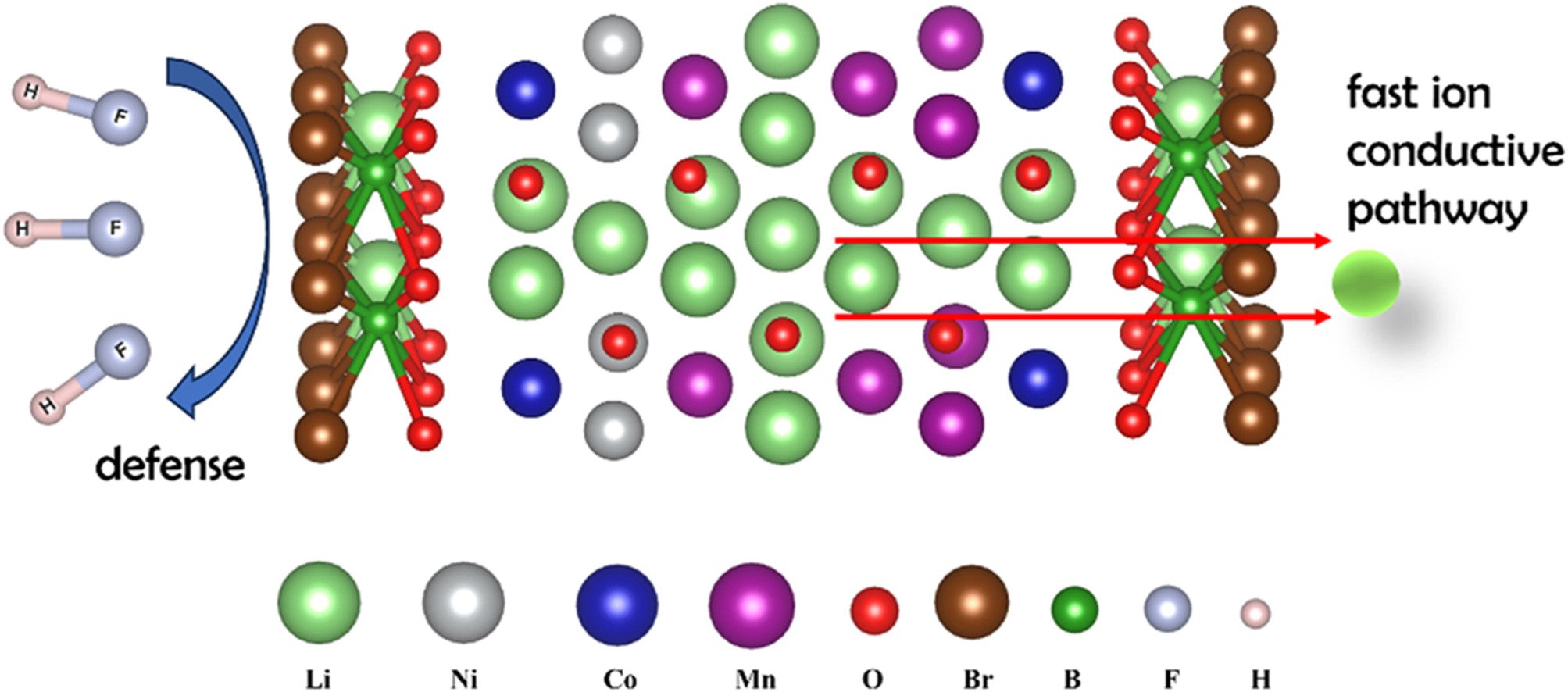
• Using first-principles and electrochemical tests, Li2O-B2O3-LiBr's effect on lithium-rich cathodes is examined.
• Li2O-B2O3-LiBr boosts lithium-rich cathodes, revealing ionic interactions and enhancing structural stability.
• Guides design optimization of lithium-rich cathode materials, potentially improving their efficiency and lifespan.
Lithium-rich cathode materials have garnered significant attention in the energy sector due to their high specific capacity. However, severe capacity degradation impedes their large-scale application. The employment of fast ion conductors for coating has shown potential in improving their electrochemical performance, yet the structural and chemical mechanisms underlying this improvement remain unclear. In this study, we systematically analyze, through first-principles calculations, the mechanism by which Li2O-B2O3-LiBr (Hereafter referred to as LBB) coating enhances the electrochemical performance of the lithium-rich layered cathode material 0.5Li2MnO3·0.5LiNi1/3Co1/3Mn1/3O2 (Hereafter referred to as OLO). Our calculations reveal that the LBB coating introduces a more negative valence charge (average −0.14 e) around the oxygen atoms surrounding transition metals, thereby strengthening metal-oxygen interactions. This interaction mitigates irreversible oxygen oxidation caused by anionic redox reactions under high voltages, reducing irreversible structural changes during battery operation. Furthermore, while the migration barrier for Li+ in OLO is 0.61 eV, the LBB coating acts as a rapid conduit during the Li+ deintercalation process, reducing the migration barrier to 0.32 eV and slightly lowering the internal migration barrier within OLO to 0.43 eV. Calculations of binding energies to electrolyte byproducts HF before and after coating (at −7.421 and −3.253 eV, respectively) demonstrate that the LBB coating effectively resists HF corrosion. Subsequent electrochemical performance studies corroborated these findings. The OLO cathode with a 2% LBB coating exhibited a discharge capacity of 157.12 mAh g−1 after 100 cycles, with a capacity retention rate of 80.38%, whereas the uncoated OLO displayed only 141.67 mAh g−1 and a 72.45% capacity retention. At a 2 C rate, with the 2 wt% LBB-coated sample maintaining a discharge capacity of 140.22 mAh g−1 compared to only 107.02 mAh g−1 for the uncoated OLO.