- Volumes 96-107 (2025)
-
Volumes 84-95 (2024)
-
Volume 95
Pages 1-392 (December 2024)
-
Volume 94
Pages 1-400 (November 2024)
-
Volume 93
Pages 1-376 (October 2024)
-
Volume 92
Pages 1-316 (September 2024)
-
Volume 91
Pages 1-378 (August 2024)
-
Volume 90
Pages 1-580 (July 2024)
-
Volume 89
Pages 1-278 (June 2024)
-
Volume 88
Pages 1-350 (May 2024)
-
Volume 87
Pages 1-338 (April 2024)
-
Volume 86
Pages 1-312 (March 2024)
-
Volume 85
Pages 1-334 (February 2024)
-
Volume 84
Pages 1-308 (January 2024)
-
Volume 95
-
Volumes 72-83 (2023)
-
Volume 83
Pages 1-258 (December 2023)
-
Volume 82
Pages 1-204 (November 2023)
-
Volume 81
Pages 1-188 (October 2023)
-
Volume 80
Pages 1-202 (September 2023)
-
Volume 79
Pages 1-172 (August 2023)
-
Volume 78
Pages 1-146 (July 2023)
-
Volume 77
Pages 1-152 (June 2023)
-
Volume 76
Pages 1-176 (May 2023)
-
Volume 75
Pages 1-228 (April 2023)
-
Volume 74
Pages 1-200 (March 2023)
-
Volume 73
Pages 1-138 (February 2023)
-
Volume 72
Pages 1-144 (January 2023)
-
Volume 83
-
Volumes 60-71 (2022)
-
Volume 71
Pages 1-108 (December 2022)
-
Volume 70
Pages 1-106 (November 2022)
-
Volume 69
Pages 1-122 (October 2022)
-
Volume 68
Pages 1-124 (September 2022)
-
Volume 67
Pages 1-102 (August 2022)
-
Volume 66
Pages 1-112 (July 2022)
-
Volume 65
Pages 1-138 (June 2022)
-
Volume 64
Pages 1-186 (May 2022)
-
Volume 63
Pages 1-124 (April 2022)
-
Volume 62
Pages 1-104 (March 2022)
-
Volume 61
Pages 1-120 (February 2022)
-
Volume 60
Pages 1-124 (January 2022)
-
Volume 71
- Volumes 54-59 (2021)
- Volumes 48-53 (2020)
- Volumes 42-47 (2019)
- Volumes 36-41 (2018)
- Volumes 30-35 (2017)
- Volumes 24-29 (2016)
- Volumes 18-23 (2015)
- Volumes 12-17 (2014)
- Volume 11 (2013)
- Volume 10 (2012)
- Volume 9 (2011)
- Volume 8 (2010)
- Volume 7 (2009)
- Volume 6 (2008)
- Volume 5 (2007)
- Volume 4 (2006)
- Volume 3 (2005)
- Volume 2 (2004)
- Volume 1 (2003)
• A new Ga-doped graphene was designed for a potential H2 storage performance using DFT method.
• This work compares adsorption energies of intrinsic and doped graphene systems for adsorption of single and multiple H2.
• Introduction of d orbital doped Ga atoms improves adsorption effect of graphene substrate on H2.
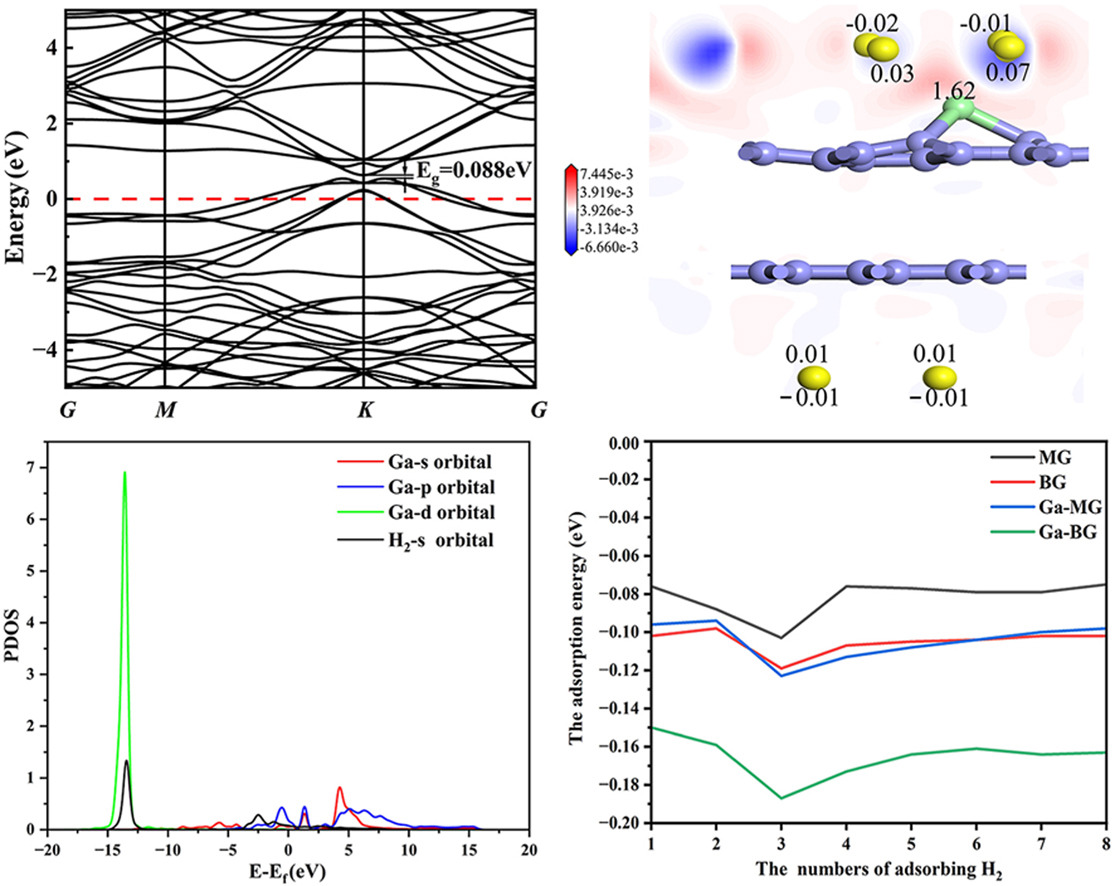
Hydrogen, as an environmentally friendly energy source, is pivotal in its storage methods for its development and effective utilization. Graphene boasts advantages such as high specific surface area, excellent electrical properties, and high tunability, making it highly promising for hydrogen storage applications. Compared to monolayer graphene, bilayer graphene exhibits a more easily controllable bandgap, showcasing its potential for hydrogen storage. Additionally, to further enhance the hydrogen adsorption capability of graphene-based substrates, doping methods are commonly employed to adjust their electrical properties. This study proposes a model for hydrogen adsorption on bilayer graphene to investigate its hydrogen storage capacity. Specifically, density functional theory (DFT) computational methods are utilized to study the adsorption of single and multiple hydrogen molecules on monolayer and bilayer graphene, with or without doping with gallium atoms. Furthermore, the underlying reasons for the enhanced hydrogen adsorption in gallium-doped bilayer graphene are systematically analyzed and elucidated. The research findings indicate that pristine graphene exhibits relatively low sensitivity to hydrogen gas, with adsorption energies of only −0.078 and −0.096 eV for monolayer graphene (MG) and bilayer graphene (BG), respectively. However, upon doping gallium atoms into MG and BG, the adsorption energy significantly increases by approximately 30.8 % and 54.1 %. For adsorbing 8 H2, with average adsorption energies reaching -0.102 eV and −0.163 eV, which is primarily due to the electron in the s orbital of H has been transferred to the d orbital of transition metal Ga. These results indicate that gallium-doped bilayer graphene holds great promise as a hydrogen storage material.