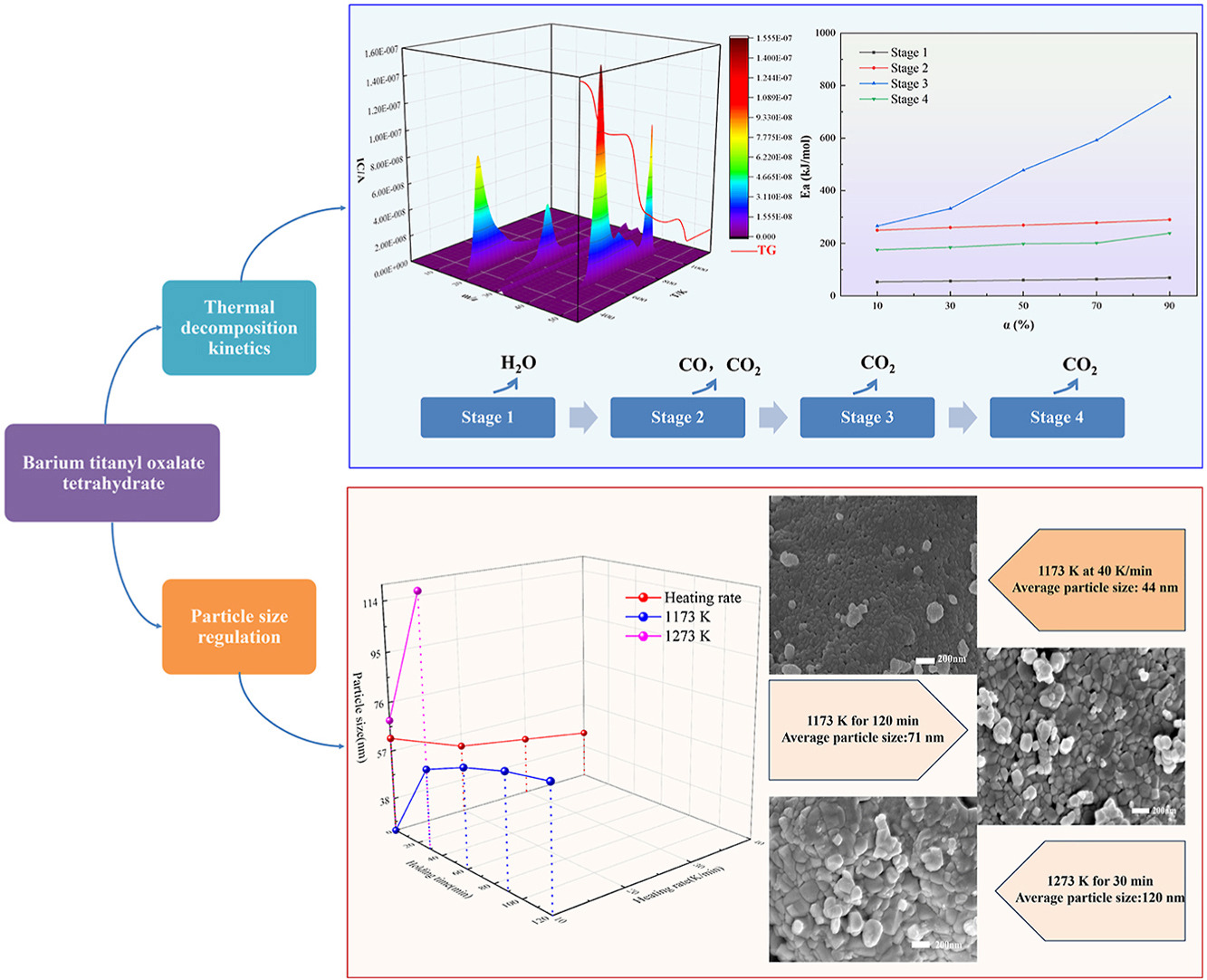
• Thermal decomposition of BTOT undergoes four distinct weight loss stages.
• KAS model is employed to calculate the average activation energy of each stage.
• Thermal decomposition mechanism and particle size regulation strategies are summarized.
• Particle size of BaTiO3 could be regulated in the range of 25–120 nm.
• Increasing the heating rate and lowering calcination temperature/time effectively inhibits BaTiO3 particle growth.
Barium titanate (BaTiO3) is an important ferroelectric and electronic ceramic material because of its outstanding dielectric and ferroelectric properties. The demand for BaTiO3 nanoparticles with adjustable particle size has increased extensively due to the miniaturization of electronic devices. The oxalate precipitation method is regarded as a highly attractive technology for fabricating BaTiO3 nanoparticles, as it enables large-scale production at a low cost. However, the calcination process is a crucial step that significantly influences the particle size and morphology of the obtained BaTiO3 nanoparticles. This study investigates the thermal decomposition mechanism and particle size regulation strategies during the calcination of barium titanyl oxalate tetrahydrate (BTOT) for fabricating BaTiO3 nanoparticles. The Kissinger-Akahira-Sunose (KAS) model is used to calculate the kinetic parameters of BTOT thermal decomposition process, and the results indicate that the decomposition process can be divided as four stages, with the average activation energy of 60.77, 269.89, 484.72, and 199.82 kJ/mol, respectively. The average activation energy reaches its maximum value in the third stage, indicating that the thermal decomposition reaction in this stage is more challenging to occur compared to the other stages. The gas release behaviors of H2O, CO, and CO2 are analyzed on-line during the thermal decomposition of BTOT, and the overall reaction mechanism is proposed. Additionally, by adjusting the calcination parameters, the particle size of BaTiO3 could be effectively regulated within the range of 25–120 nm. Increasing the heating rate from 10 to 40 K/min decreases the average particle size of BaTiO3 from 62 to 44 nm. Extending the calcination time from 0 to 120 min increases the average particle size from 25 to 71 nm. Raising the terminal temperature from 1173 to 1273 K significantly increases the average particle size from 56 to 120 nm. Briefly, accelerating the heating rate, reducing the calcination time, and lowering the calcination temperature facilitate the fabrication of BaTiO3 with a smaller particle size and more uniform morphology. This study offers a robust theoretical framework and technical guidance for optimizing the process conditions for fabricating BaTiO3 nanoparticles via controlled thermal decomposition of BTOT, while also contributing to the advancement of related technological fields.