• Pore wetting percentage of particles was improved by the oxygen functional groups.
• Potential of particles by n-hexadecane and NaOL solution was positive and negative.
• Amphiphilic reagent NaOL was more stable adsorbed to particles than n-hexadecane.
• Hydrophobicity of particles adsorbed by NaOL was larger than that by n-hexadecane.
• Porous carbon particles wetted by NaOL solution had a better floatability.
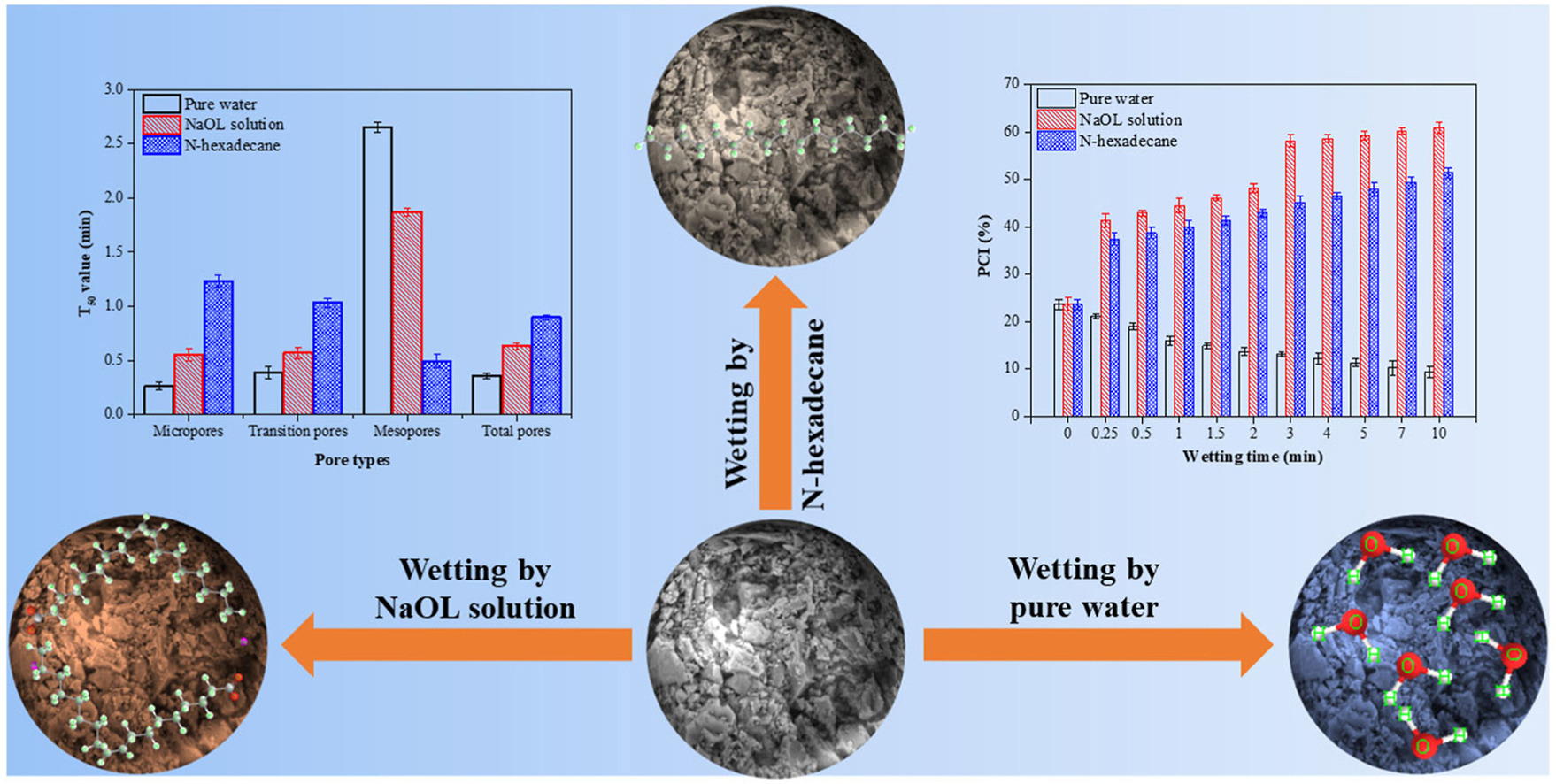
Dynamic pore wetting plays a major role in the flotation of porous mineral particles. However, the role of chemical properties of the liquids in dynamic pore wetting is not systematically studied. This paper studied the dynamic pore wetting process of the porous carbon particles by 10 g/L NaOL solution and n-hexadecane at different wetting times using a 1H low-field nuclear magnetic resonance (LF-NMR). The mechanism of chemical groups of liquids affecting the pore wetting percentage and floatability of porous particles was revealed by FTIR, zeta potential, contact angle, and particle-bubble attachment tests. The results displayed that the pore wetting percentage of the particles wetted by NaOL solution was larger than that by n-hexadecane except for mesopores. Compared with the raw sample, the surface potential of the particles wetted by n-hexadecane was decreased and that by NaOL solution were negative. The NaOL, as an amphiphilic reagent contained some hydrophobic and hydrophilic functional groups, which was different from the n-hexadecane. The NaOL molecule could be easily adsorbed into the particles by electrostatic interaction compared with n-hexadecane, achieving a better surface hydrophobicity of the particles. Thus, the floatability of porous carbon particles wetted by NaOL solution was larger than that by n-hexadecane.