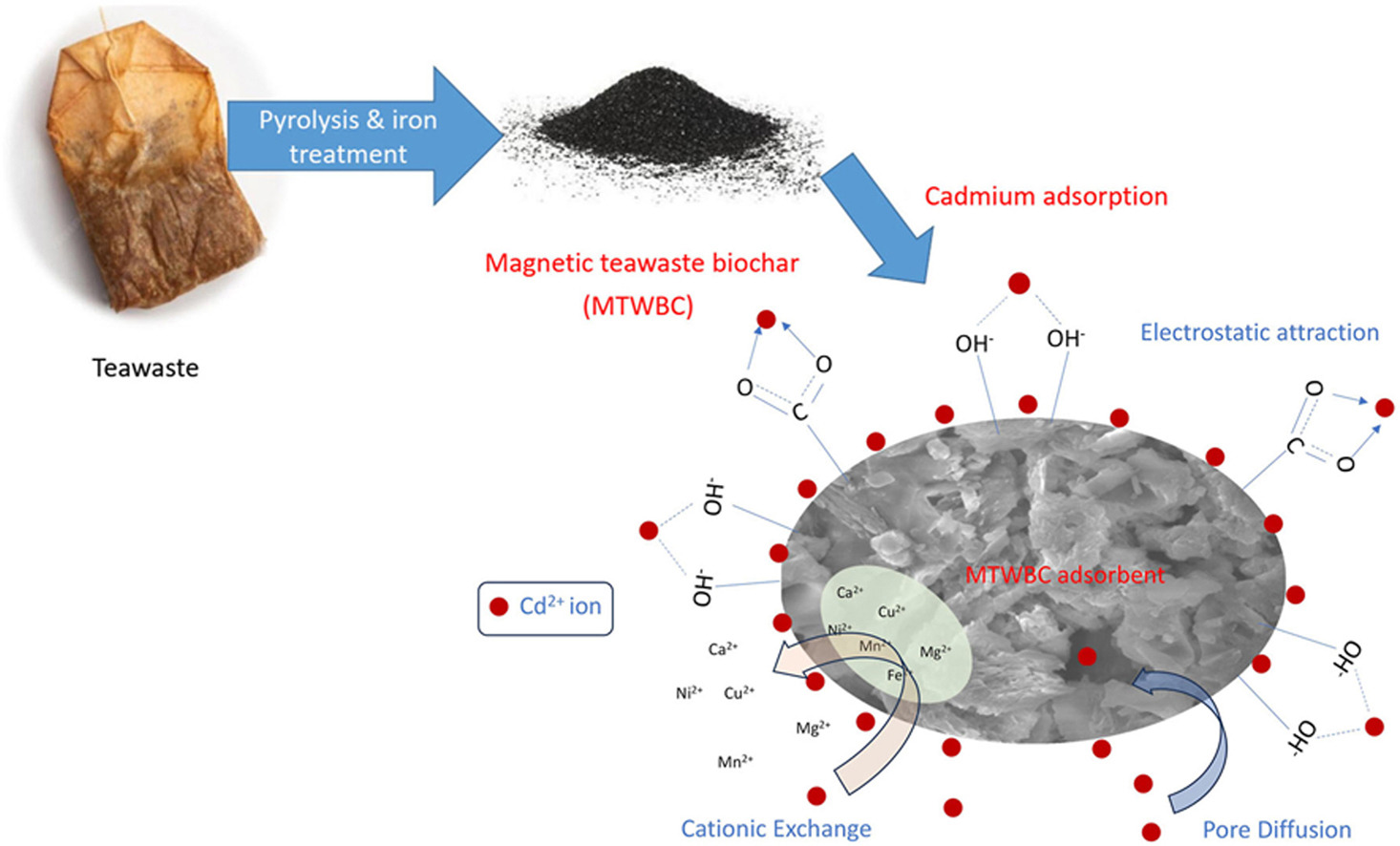
• Magnetic teawaste biochar (MTWBC) was successfully produced from teawaste.
• Optimum iron nanoparticles loading and pyrolysis temperature was determined.
• Magnetic teawaste biochar produced has high removal capacity towards Cd(II) (∼7.23 mmol/g).
• Cd(II) removal process was found to be endothermic.
The Response Surface Methodology (RSM), specifically the face-centred central design, was employed to determine the optimal pyrolysis conditions for producing magnetic biochar from teawaste with the highest capacity for removing cadmium (Cd2+) ions. Several process conditions were investigated, including pyrolysis temperature (200, 350, 500 °C), duration of pyrolysis (2, 3, 4 h), and the concentration of iron chloride added (0.02, 0.05, 0.09 g/g). The sample obtained through pyrolysis at a temperature of 200 °C for a duration of 4 h with an iron chloride concentration of 0.09 g/g exhibited the highest removal efficiency of 80% for Cd2+. Results show that the magnetic teawaste biochar (MTWBC) possessed a maximum Langmuir capacity of 7.23 mmol/g at 298 K. The experimental data obtained for the three temperatures studied were best described by the Freundlich isotherm model. As for the kinetics of the experiment, the Modified Freundlich Kinetic model provided the most accurate fit. The Fourier Transform Infrared (FT-IR) analysis conducted on the MTWBC samples, both before and after adsorption, demonstrated that the removal of Cd2+ by MTWBC involved chemisorption, which engaged specific functional groups present on the surface of MTWBC. Overall, these results highlight the promising potential of MTWBC as an economically viable bio-adsorbent for the removal of cadmium from contaminated water sources.