• Hollow Zn-BTC was obtained through controlling hydrothermal reaction time.
• Hollow Zn-BTC exhibits high adsorption capacity and fast diffusion rate toward thiophene.
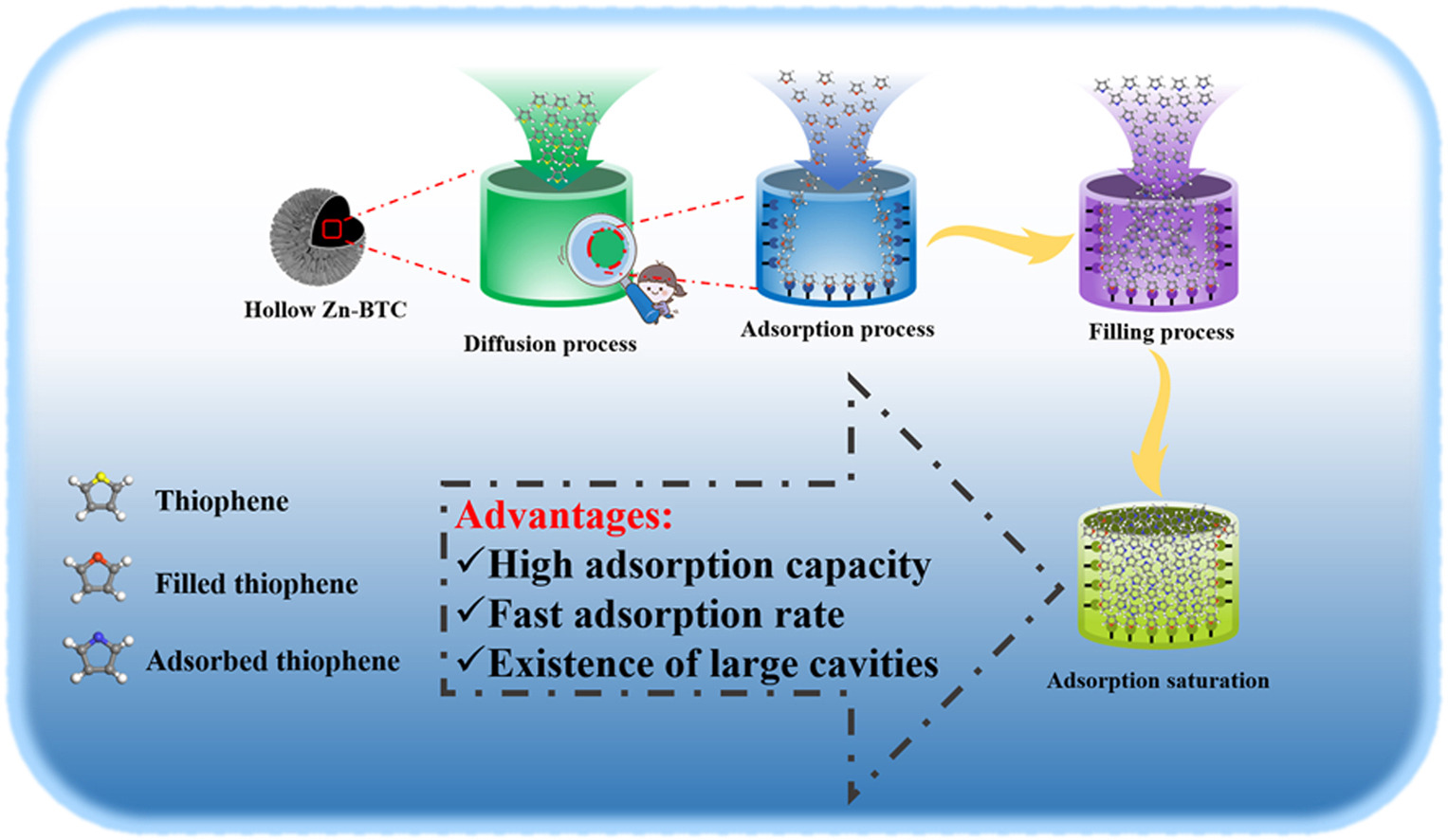
• Pore-filling effect significantly enhances the adsorption performance of thiophene.
Conventional metal-organic frameworks (MOFs) have potential applications in adsorption desulfurization due to their open metal sites, structural diversity, and high specific surface area. However, the narrow internal pores limit the adsorption and diffusion of thiophene sulfur molecules (TSM). Therefore, we constructed a novel hollow-type Zn-BTC (HZB) adsorbent by a self-assembled hydrothermal method without using dopant templating agents. The presence of a large internal cavity can be seen by SEM. The results of batch tests showed that the hollow structure Zn-BTC has higher sulfur absorption capacity and faster diffusion rate. The adsorption isotherms were in good agreement with both Freundlich and Dubinin-Radushkevich (D-R) models, suggesting multilayer adsorption combined with pore-filling effect. The maximum adsorption capacity of the TSMs could reach 80% within 30 min, which enabled rapid adsorption in accordance with the pseudo-second-order kinetic model. The adsorption mechanism involves pore filling, ligand effect, and π-π attraction. In addition, HZB-3 maintained good adsorption capacity after the fifth cycle, showing good reusability and stability. This work provides a new strategy for TMS capture using hollow MOF.