- Volumes 108-119 (2025)
-
Volumes 96-107 (2025)
-
Volume 107
Pages 1-376 (December 2025)
-
Volume 106
Pages 1-336 (November 2025)
-
Volume 105
Pages 1-356 (October 2025)
-
Volume 104
Pages 1-332 (September 2025)
-
Volume 103
Pages 1-314 (August 2025)
-
Volume 102
Pages 1-276 (July 2025)
-
Volume 101
Pages 1-166 (June 2025)
-
Volume 100
Pages 1-256 (May 2025)
-
Volume 99
Pages 1-242 (April 2025)
-
Volume 98
Pages 1-288 (March 2025)
-
Volume 97
Pages 1-256 (February 2025)
-
Volume 96
Pages 1-340 (January 2025)
-
Volume 107
-
Volumes 84-95 (2024)
-
Volume 95
Pages 1-392 (December 2024)
-
Volume 94
Pages 1-400 (November 2024)
-
Volume 93
Pages 1-376 (October 2024)
-
Volume 92
Pages 1-316 (September 2024)
-
Volume 91
Pages 1-378 (August 2024)
-
Volume 90
Pages 1-580 (July 2024)
-
Volume 89
Pages 1-278 (June 2024)
-
Volume 88
Pages 1-350 (May 2024)
-
Volume 87
Pages 1-338 (April 2024)
-
Volume 86
Pages 1-312 (March 2024)
-
Volume 85
Pages 1-334 (February 2024)
-
Volume 84
Pages 1-308 (January 2024)
-
Volume 95
-
Volumes 72-83 (2023)
-
Volume 83
Pages 1-258 (December 2023)
-
Volume 82
Pages 1-204 (November 2023)
-
Volume 81
Pages 1-188 (October 2023)
-
Volume 80
Pages 1-202 (September 2023)
-
Volume 79
Pages 1-172 (August 2023)
-
Volume 78
Pages 1-146 (July 2023)
-
Volume 77
Pages 1-152 (June 2023)
-
Volume 76
Pages 1-176 (May 2023)
-
Volume 75
Pages 1-228 (April 2023)
-
Volume 74
Pages 1-200 (March 2023)
-
Volume 73
Pages 1-138 (February 2023)
-
Volume 72
Pages 1-144 (January 2023)
-
Volume 83
-
Volumes 60-71 (2022)
-
Volume 71
Pages 1-108 (December 2022)
-
Volume 70
Pages 1-106 (November 2022)
-
Volume 69
Pages 1-122 (October 2022)
-
Volume 68
Pages 1-124 (September 2022)
-
Volume 67
Pages 1-102 (August 2022)
-
Volume 66
Pages 1-112 (July 2022)
-
Volume 65
Pages 1-138 (June 2022)
-
Volume 64
Pages 1-186 (May 2022)
-
Volume 63
Pages 1-124 (April 2022)
-
Volume 62
Pages 1-104 (March 2022)
-
Volume 61
Pages 1-120 (February 2022)
-
Volume 60
Pages 1-124 (January 2022)
-
Volume 71
- Volumes 54-59 (2021)
- Volumes 48-53 (2020)
- Volumes 42-47 (2019)
- Volumes 36-41 (2018)
- Volumes 30-35 (2017)
- Volumes 24-29 (2016)
- Volumes 18-23 (2015)
- Volumes 12-17 (2014)
- Volume 11 (2013)
- Volume 10 (2012)
- Volume 9 (2011)
- Volume 8 (2010)
- Volume 7 (2009)
- Volume 6 (2008)
- Volume 5 (2007)
- Volume 4 (2006)
- Volume 3 (2005)
- Volume 2 (2004)
- Volume 1 (2003)
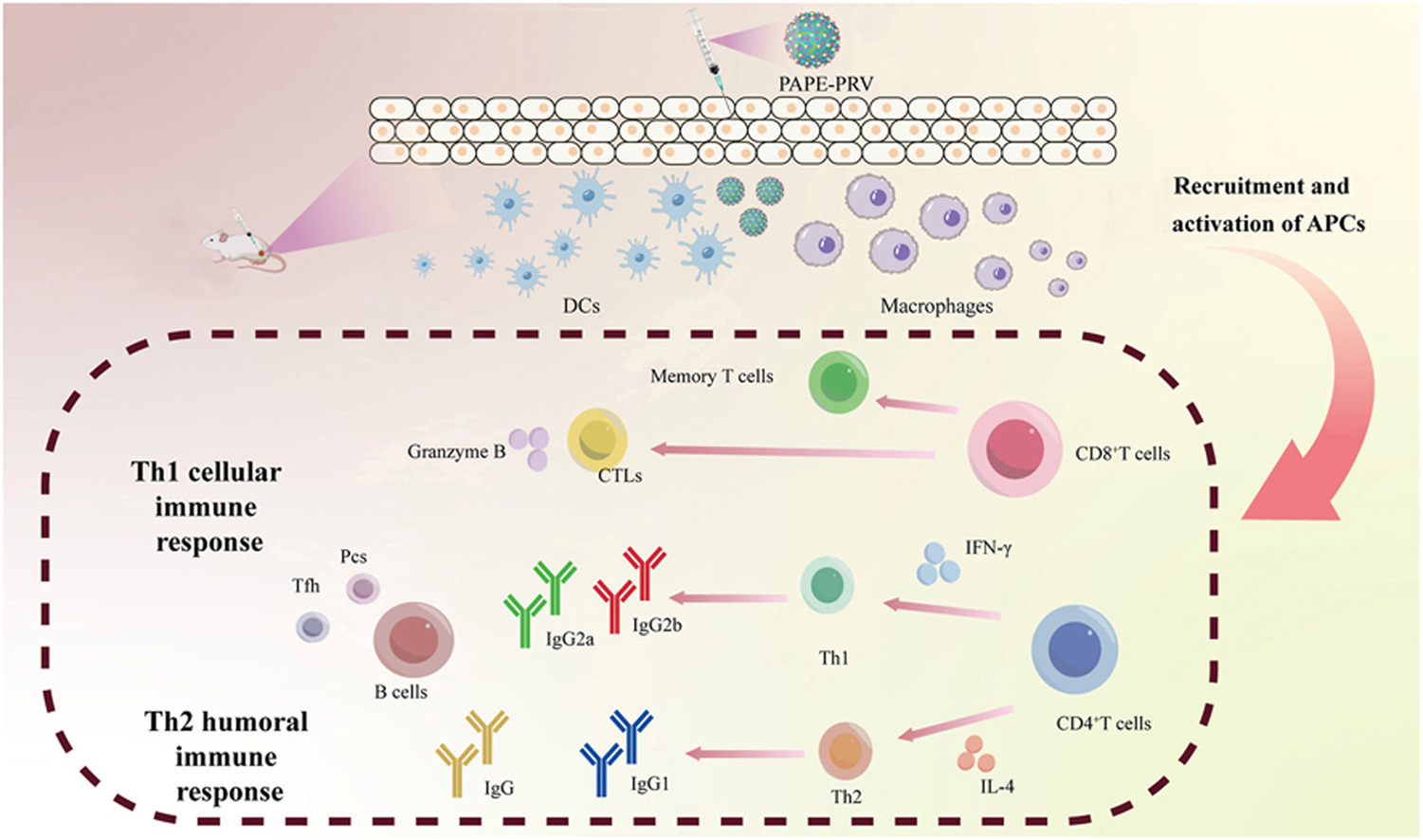
• PAPE-PRV increased the recruitment and activation of APCs at the injection site.
• PAPE-PRV provoked strong cellular and humoral immune responses.
• PAPE compensated for the inadequacy of alum in stimulating cellular immune responses.
• Compared to ISA206 adjuvant, PAPE-PRV induced stronger cellular immune responses.
Vaccination with inactivated porcine pseudorabies virus (PRV) vaccines is a common strategy for the prevention of PRV infection. However, due to the insufficient immunogenicity, the protective efficacy of inactivated vaccines remains deficient. Consequently, there is an urgent need to develop potent adjuvants to enhance the effectiveness of inactivated PRV vaccines. In previous study, we successfully developed a novel vaccine adjuvant delivery system, which the Poria cocos polysaccharide-loaded Alhydrogel was employed as colloidal stabilizers, and squalene was utilized as the oil phase to form stable Pickering emulsions (PAPE). The PAPE combined the immunostimulatory effects of Poria cocos polysaccharide, the inherent immunostimulant properties of the Alhydrogel adjuvant, and the characteristics of the Pickering emulsions delivery system. Herein, we found that PAPE has the potential to function as a delivery system to promote antigen internalization by macrophages via scavenger receptor A-mediated endocytosis. PAPE compensated for the inadequacy of alum adjuvants in efficiently stimulating cell-mediated immune responses. As the adjuvant for inactivated PRV vaccine, PAPE increased the recruitment and activation of antigen-presenting cells at the injection site, and provoked strong cellular and humoral immune responses. Notably, compared to MONTANIDE ISA206 adjuvant, PAPE markedly improved the induction of CD4+ and CD8+ T cells, the activation of CD8+ cytotoxic T lymphocytes, the production of IFN-γ, and the response of memory CD8+ T cells, thereby inducing a stronger cellular immune response. Our findings highlight the efficacy of PAPE as an adjuvant for PRV vaccines, offering new insights for the development of veterinary vaccines.