- Volumes 108-119 (2025)
-
Volumes 96-107 (2025)
-
Volume 107
Pages 1-376 (December 2025)
-
Volume 106
Pages 1-336 (November 2025)
-
Volume 105
Pages 1-356 (October 2025)
-
Volume 104
Pages 1-332 (September 2025)
-
Volume 103
Pages 1-314 (August 2025)
-
Volume 102
Pages 1-276 (July 2025)
-
Volume 101
Pages 1-166 (June 2025)
-
Volume 100
Pages 1-256 (May 2025)
-
Volume 99
Pages 1-242 (April 2025)
-
Volume 98
Pages 1-288 (March 2025)
-
Volume 97
Pages 1-256 (February 2025)
-
Volume 96
Pages 1-340 (January 2025)
-
Volume 107
-
Volumes 84-95 (2024)
-
Volume 95
Pages 1-392 (December 2024)
-
Volume 94
Pages 1-400 (November 2024)
-
Volume 93
Pages 1-376 (October 2024)
-
Volume 92
Pages 1-316 (September 2024)
-
Volume 91
Pages 1-378 (August 2024)
-
Volume 90
Pages 1-580 (July 2024)
-
Volume 89
Pages 1-278 (June 2024)
-
Volume 88
Pages 1-350 (May 2024)
-
Volume 87
Pages 1-338 (April 2024)
-
Volume 86
Pages 1-312 (March 2024)
-
Volume 85
Pages 1-334 (February 2024)
-
Volume 84
Pages 1-308 (January 2024)
-
Volume 95
-
Volumes 72-83 (2023)
-
Volume 83
Pages 1-258 (December 2023)
-
Volume 82
Pages 1-204 (November 2023)
-
Volume 81
Pages 1-188 (October 2023)
-
Volume 80
Pages 1-202 (September 2023)
-
Volume 79
Pages 1-172 (August 2023)
-
Volume 78
Pages 1-146 (July 2023)
-
Volume 77
Pages 1-152 (June 2023)
-
Volume 76
Pages 1-176 (May 2023)
-
Volume 75
Pages 1-228 (April 2023)
-
Volume 74
Pages 1-200 (March 2023)
-
Volume 73
Pages 1-138 (February 2023)
-
Volume 72
Pages 1-144 (January 2023)
-
Volume 83
-
Volumes 60-71 (2022)
-
Volume 71
Pages 1-108 (December 2022)
-
Volume 70
Pages 1-106 (November 2022)
-
Volume 69
Pages 1-122 (October 2022)
-
Volume 68
Pages 1-124 (September 2022)
-
Volume 67
Pages 1-102 (August 2022)
-
Volume 66
Pages 1-112 (July 2022)
-
Volume 65
Pages 1-138 (June 2022)
-
Volume 64
Pages 1-186 (May 2022)
-
Volume 63
Pages 1-124 (April 2022)
-
Volume 62
Pages 1-104 (March 2022)
-
Volume 61
Pages 1-120 (February 2022)
-
Volume 60
Pages 1-124 (January 2022)
-
Volume 71
- Volumes 54-59 (2021)
- Volumes 48-53 (2020)
- Volumes 42-47 (2019)
- Volumes 36-41 (2018)
- Volumes 30-35 (2017)
- Volumes 24-29 (2016)
- Volumes 18-23 (2015)
- Volumes 12-17 (2014)
- Volume 11 (2013)
- Volume 10 (2012)
- Volume 9 (2011)
- Volume 8 (2010)
- Volume 7 (2009)
- Volume 6 (2008)
- Volume 5 (2007)
- Volume 4 (2006)
- Volume 3 (2005)
- Volume 2 (2004)
- Volume 1 (2003)
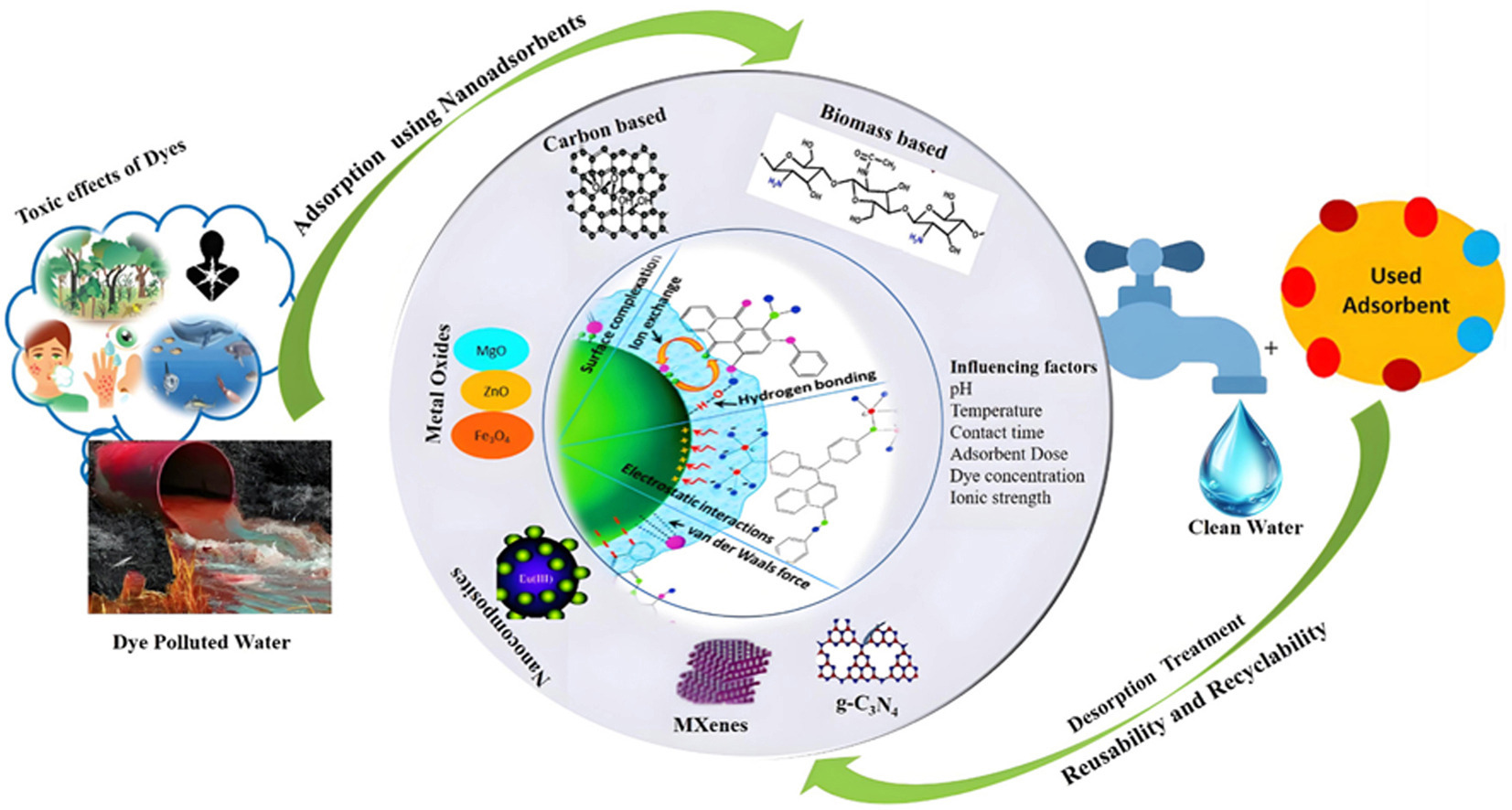
• Reviews major nanoadsorbents (metal oxides, carbon nanostructures, biomass-derived nanomaterials, MXenes and g-C3N4) with 2-10 times higher dye uptake.
• Elucidates dominant dye adsorption mechanisms and effects of pH, temperature, contact time, adsorbent dose, dye concentration and co-ions.
• Evaluates regeneration and reusability, showing75-90%adsorption efficiencies retained over repeated cycles.
• Highlights scalability, stability, environmental safety challenges and future directions for industrial-scale application.
Industrial effluents are the major source of persistent and non-biodegradable synthetic dyes that pose serious health and environmental threats. Among the various treatment methods, adsorption has emerged as a simple, cost-effective and highly efficient (>90%) approach for dye removal from wastewater. In this context, nanoadsorbents have gained considerable attention due to large surface areas (10-1000 m2 g-1), tunable porosity, functionalized surfaces enabling the adsorption capacities 2-10 times higher than those of conventional adsorbents. A wide range of nanoadsorbents including metal and metal oxide nanoparticles (NPs) (MgO, TiO2, Fe3O4, ZnO etc.), carbon-based nanostructures (graphene oxide, carbon nanotubes etc.), biomass driven nano sized entities and emerging nanoadsorbents such as MXenes, g-C3N4 have been explored for efficient removal of both cationic and anionic dyes. Dye adsorption is governed by mechanisms such as electrostatic interactions, hydrogen bonding, π-π stacking, and surface complexation which are strongly influenced by pH, temperature, initial dye concentration and adsorbent dose. Importantly, regeneration studies indicate that many nanoadsorbents retain 75–90% of their initial efficiency over the multiple cycles highlighting their reusability and economic potential. This review critically summarizes recent advancements in nanoadsorbent-based dye removal with emphasis on adsorption mechanism, process parameters, regeneration performance along with addressing the key challenges related to scalability, long term stability, and potential environmental risks that must be resolved for sustainable wastewater treatment.